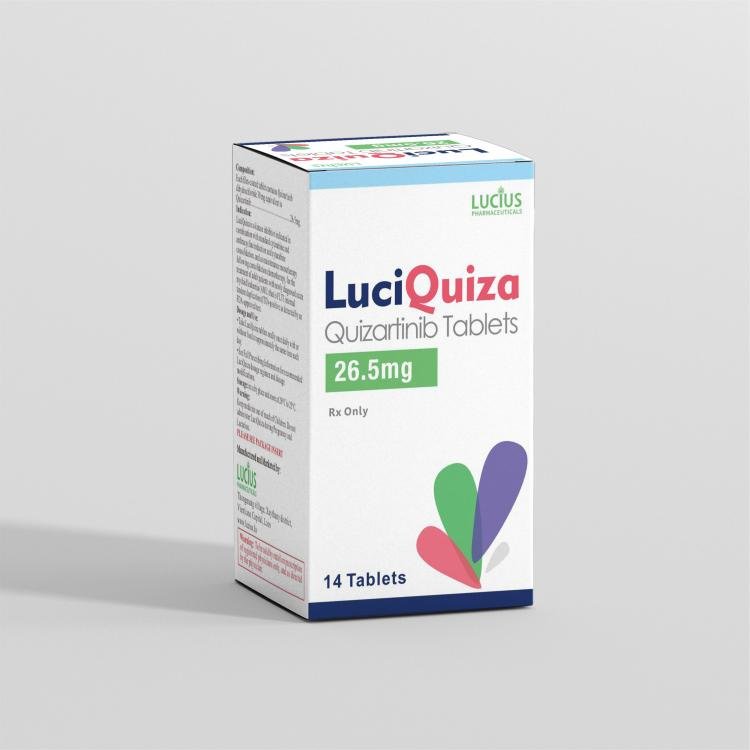
Product Name: LuciQuiza
Manufacturer: Lucius Pharmaceuticals (Laos) Co., Ltd.
English Name: Quizartinib Tablets
Approval Number: 06 L 1106/24 (26.5mg) Indications LuciQuiza is a kinase inhibitor indicated, in combination with standard cytarabine and anthracycline induction and cytarabine consolidation chemotherapy, followed by maintenance monotherapy with LuciQuiza after consolidation chemotherapy, for the treatment of adult patients with newly diagnosed acute myeloid leukemia (AML) harboring FLT3 internal tandem duplication (ITD) mutations, as detected by an FDA-approved test. Use Limitation:
LuciQuiza is not indicated as maintenance monotherapy following allogeneic hematopoietic stem cell transplantation (HSCT); benefit in improving overall survival has not been demonstrated in this setting. Dosage and Administration
Manufacturer: Lucius Pharmaceuticals (Laos) Co., Ltd.
English Name: Quizartinib Tablets
Approval Number: 06 L 1106/24 (26.5mg) Indications LuciQuiza is a kinase inhibitor indicated, in combination with standard cytarabine and anthracycline induction and cytarabine consolidation chemotherapy, followed by maintenance monotherapy with LuciQuiza after consolidation chemotherapy, for the treatment of adult patients with newly diagnosed acute myeloid leukemia (AML) harboring FLT3 internal tandem duplication (ITD) mutations, as detected by an FDA-approved test. Use Limitation:
LuciQuiza is not indicated as maintenance monotherapy following allogeneic hematopoietic stem cell transplantation (HSCT); benefit in improving overall survival has not been demonstrated in this setting. Dosage and Administration
- Take LuciQuiza tablets orally once daily at approximately the same time each day, with or without food.
- Refer to the full prescribing information for the recommended dosing regimen and dose modifications.
Specification
26.5 mg × 14 tablets per box.

Reviews
There are no reviews yet.