Product Details
Product Information
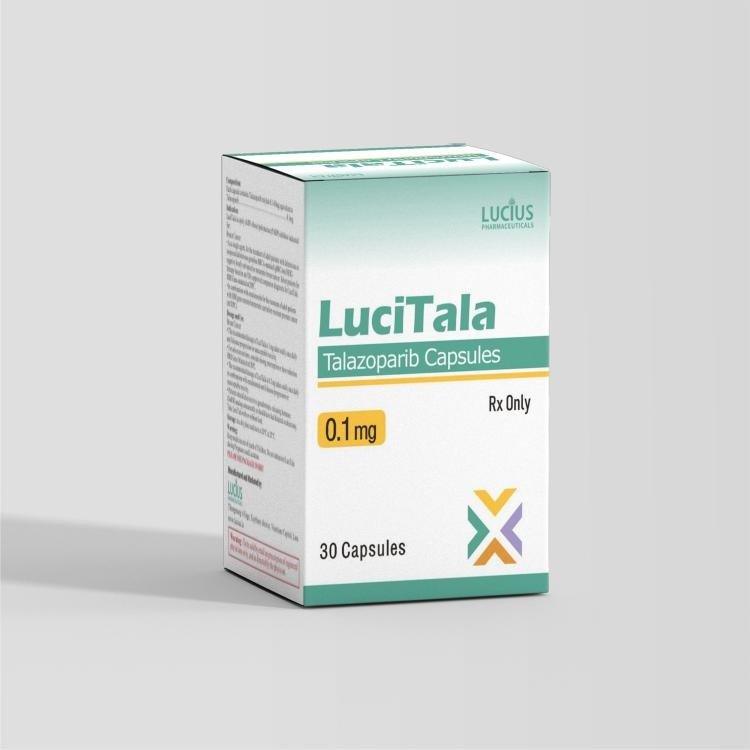
- Product name: LuciTala Talazopanib Talazoparib 0.1mg
- Manufacturer / brand: Laos Lucius Pharma
- Active ingredient: Talazoparib
- Current strength: 0.1mg
- SKU: LU-ITEM-154
- Site category: BRCA target, HRR target, other cancer
Product Summary
LuciTala Talazopanib Talazoparib 0.1mg is an AISTIKA-listed product supplied by Laos Lucius Pharma. This page summarizes the product identity, strength, SKU, site category, and public medical reference information. Product name, manufacturer, packaging, and strength follow this AISTIKA product page.
Active Ingredient and Reference Data
Talazoparib is the active ingredient used for this product page. Public prescribing information for Talazoparib was used for the medical reference sections. Medical details are provided for reference and must be interpreted by a qualified healthcare professional.
Mechanism of Action
Talazoparib is an inhibitor of PARP enzymes, including PARP1 and PARP2, which play a role in DNA repair. Talazoparib anti-tumor activity was observed in patient-derived xenograft breast cancer models bearing mutated BRCA1 or mutated BRCA2 or wild type BRCA1 and BRCA2 .
Reference Indications
Talazoparib is a poly (ADP-ribose) polymerase (PARP) inhibitor indicated for: Breast Cancer As a single agent, for the treatment of adult patients with deleterious or suspected deleterious germline BRCA -mutated (g BRCA m) HER2-negative locally advanced or metastatic breast cancer. Select patients for therapy based on an FDA-approved companion diagnostic for Talazoparib. HRR Gene-mutated Metastatic Castration-Resistant Prostate Cancer (mCRPC) In combination with enzalutamide for the treatment of adult patients with HRR gene-mutated metastatic castration-resistant prostate cancer (mCRPC). Select patients for therapy based on an FDA-approved companion diagnostic for Talazoparib .
Reference Dosage and Administration
Recommended dosage of Talazoparib is 1 mg taken orally once daily until disease progression or unacceptable toxicity. Take Talazoparib with or without food.
With this 0.1mg product, that corresponds to 10 x 0.1mg tablets when that reference dose is clinically appropriate.
This reference dosage is not an individualized prescription. Dose changes, treatment interruptions, or discontinuation must be directed by a physician.
Important Safety Information
Myelodysplastic Syndrome/Acute Myeloid Leukemia (MDS/AML) : MDS/AML occurred in patients exposed to Talazoparib, and some cases were fatal. Monitor patients for hematological toxicity and discontinue if MDS/AML is confirmed. Myelosuppression : Talazoparib may affect hematopoiesis and can cause anemia, neutropenia, and/or thrombocytopenia. Embryo-Fetal Toxicity : Talazoparib can cause fetal harm. Advise of the potential risk to the fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 ) 5.1 Myelodysplastic Syndrome/Acute Myeloid Leukemia Myelodysplastic Syndrome/Acute Myeloid Leukemia (MDS/AML), including cases with a fatal outcome, has been reported in patients who received Talazoparib. Overall, MDS/AML has been reported in 0.4% (3 out of 788) of solid tumor patients treated with Talazoparib as a single agent in clinical studies.
Common Adverse Reactions and Monitoring
The data described in the section reflect exposure to single agent Talazoparib in solid tumor clinical studies, including 286 patients enrolled in EMBRACA trial and to Talazoparib 0.5 mg daily with enzalutamide in 511 patients enrolled in the TALAPRO-2 trial that included 197 patients with HRR gene-mutated mCRPC. g BRCA m HER2-negative Locally Advanced or Metastatic Breast Cancer EMBRACA The safety of Talazoparib as a single agent was evaluated in g BRCA m patients with HER2-negative locally advanced or metastatic breast cancer who had previously received no more than 3 lines of chemotherapy for the treatment of locally advanced/metastatic disease .
Drug Interactions and Special Populations
P-gp Inhibitors : Reduce the dose when coadministered with certain P-gp inhibitors. If coadministration of Talazoparib with these P-gp inhibitors cannot be avoided, reduce the dose of Talazoparib . When the P-gp inhibitor is discontinued, increase the dose of Talazoparib . HRR Gene-mutated mCRPC The effect of coadministration of P-gp inhibitors on talazoparib exposure when Talazoparib is taken with enzalutamide has not been studied. Lactation : Advise women not to breastfeed. ( 2.6 , 8.7 ) 8.1 Pregnancy Risk Summary Based on findings from animal studies and its mechanism of action , Talazoparib can cause embryo-fetal harm when administered to a pregnant woman. There are no available data on Talazoparib use in pregnant women to inform a drug-associated risk.
Storage and Purchase Notes
Store according to the package or pharmacist instructions and keep out of reach of children. Before ordering, confirm product name, strength, quantity, price, shipping details, and payment method. After receipt, check packaging, batch number, expiration date, and storage conditions.
Sources
Sources: Talzenna public prescribing information; openFDA/DailyMed public label data. These sources are used for public medical reference. Product information follows this AISTIKA product page.
DisclaimerThis page is for product information and public-label reference only. It does not provide diagnosis, prescription, or individualized medical advice. Consult a physician for treatment decisions.



Reviews
There are no reviews yet.